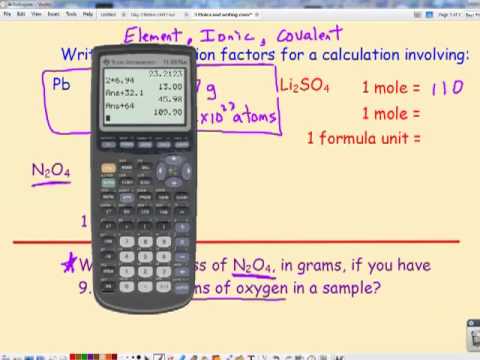
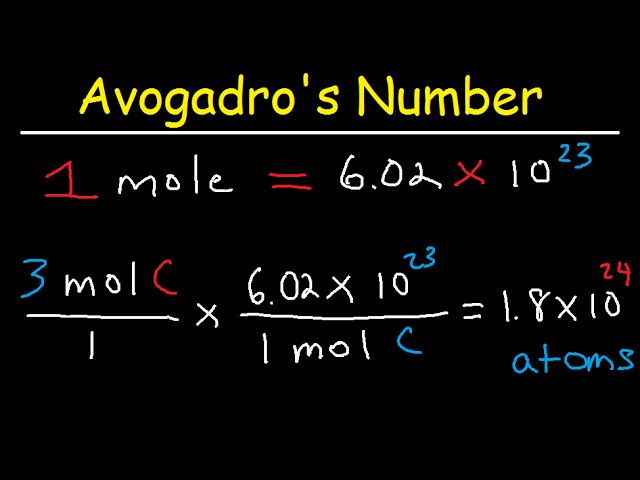Clip of M1.3: The Mole Part 1 (Avogadro's Number) - SchoolTube - Safe video sharing and management for K12

1 CALCULATOR USE FOR SCIENTIFIC NOTATION PUT INTO SCIENTIFIC NOTATION MODE TO GET ANSWERS IN SCI NOT PUT INTO NORMAL MODE TO GET DECIMAL NUMBERS. - ppt download

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5

Use Avogadro's Number to Calculate the Mass of a Single Atom | Relative atomic mass, Chemistry, Atom
Is there a preset function in Texas Instrument-84 Plus calculators for Avogadro's number? Can you add one? - Quora

Objective: To reintroduce the mole and introduce two mole conversions Do Now: Solve each proportion = x2. 10 = x. - ppt download

Section 2 - Avogadro's Number and the Mole - Part 1 | Math Tutor DVD - Online Math Help, Math Homework Help, Math Problems, Math Practice!

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5