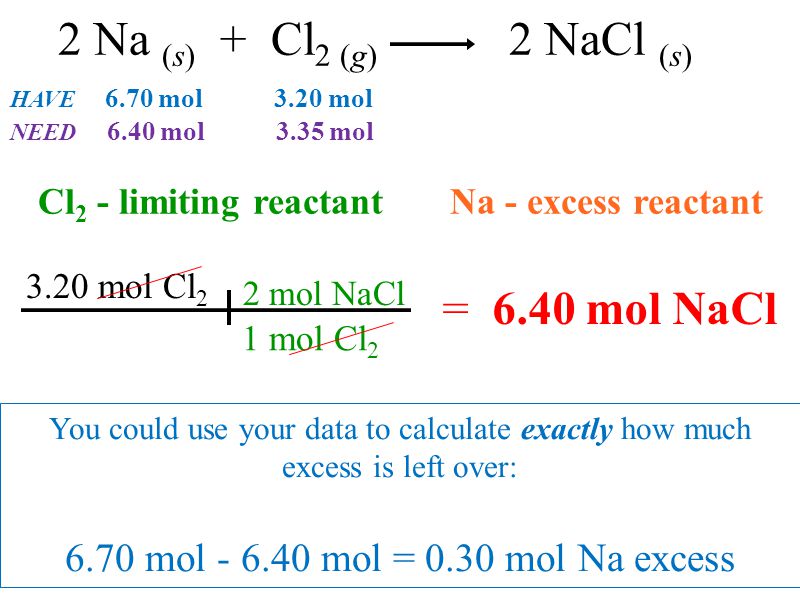
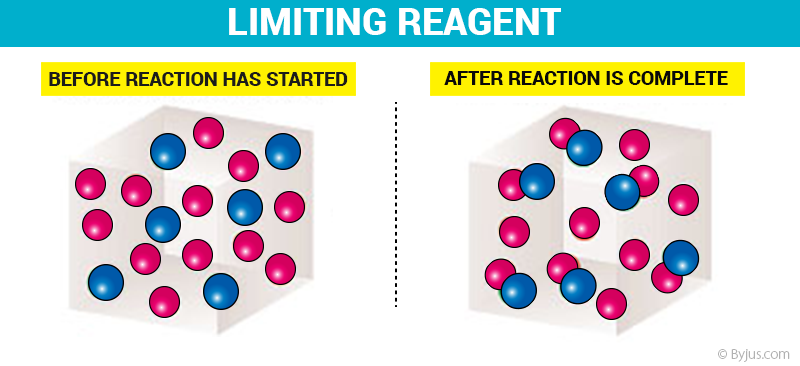
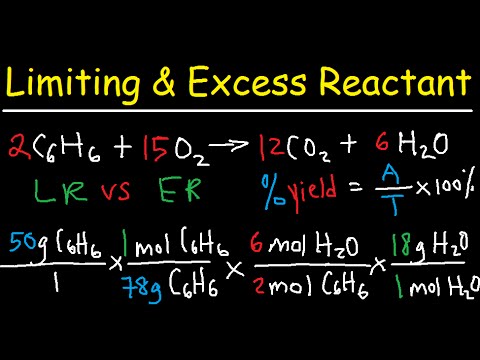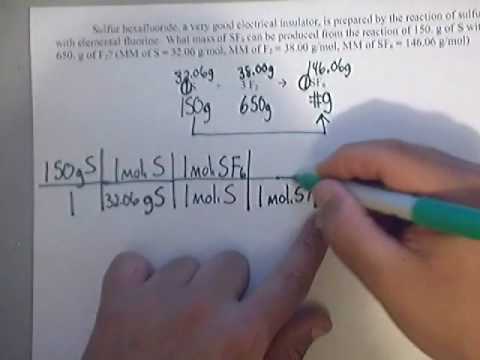Friday March 27 Objective: Determine the limiting reactant in a chemical reaction. Determine how much product can be made. Checkpoint: Given the equation: - ppt download
![Grade 11: Stoichiometry] Is there not a need to find out the limiting reagent when we calculate the mass of ammonia? : r/HomeworkHelp Grade 11: Stoichiometry] Is there not a need to find out the limiting reagent when we calculate the mass of ammonia? : r/HomeworkHelp](https://i.redd.it/4abku36f34j51.jpg)
Grade 11: Stoichiometry] Is there not a need to find out the limiting reagent when we calculate the mass of ammonia? : r/HomeworkHelp

How to Find How Much Excess Reactant Remains Examples, Practice Problems, Questions, Summary - YouTube

CHEM 101: Dimensional Analysis Limiting Reagent, Theoretical Yield, Percent Yield, Excess Reactant 2 - YouTube

Limiting Reactants & Excess. Limiting Reactant Calculations In many chemical reactions an excess of one reactant is added to ensure complete reaction. - ppt download