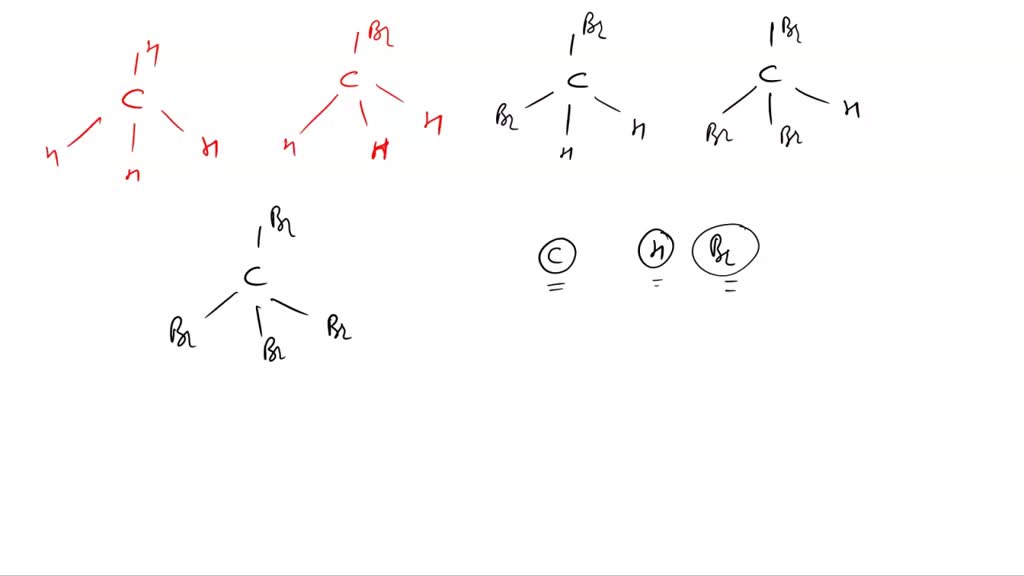
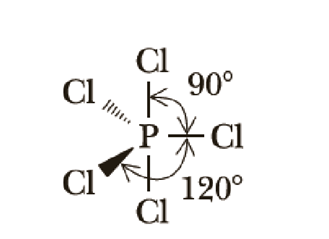
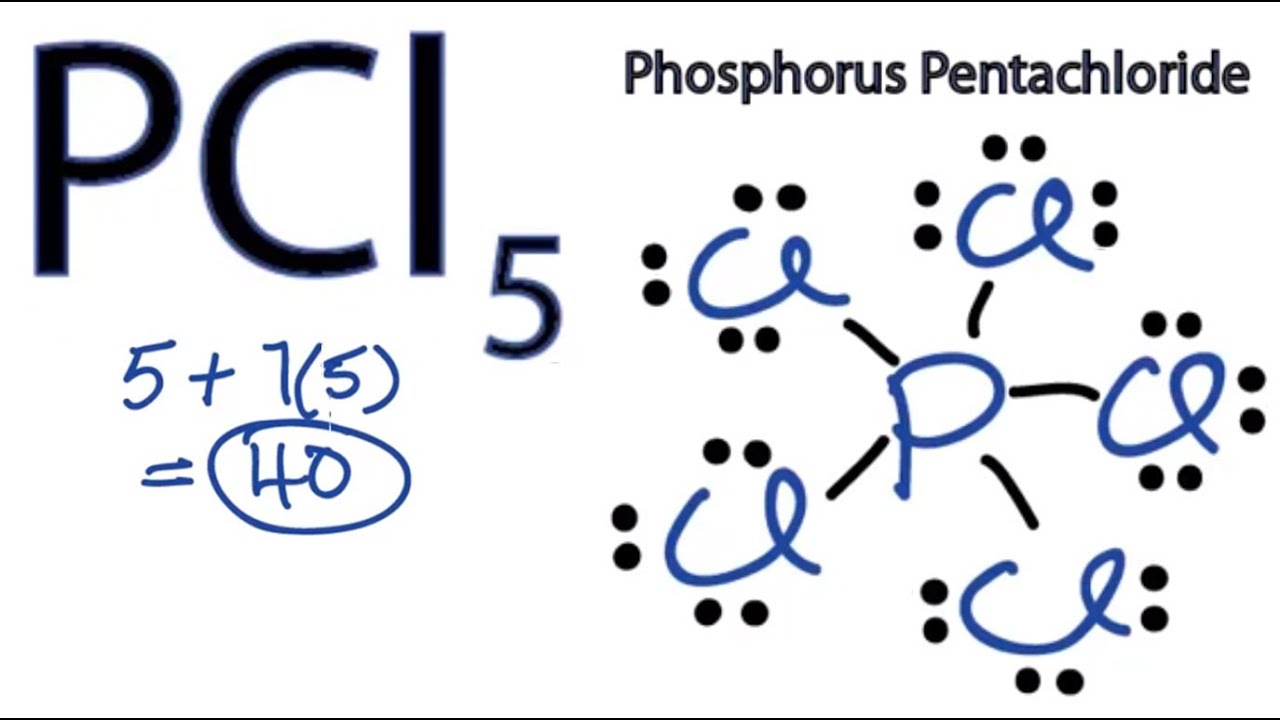
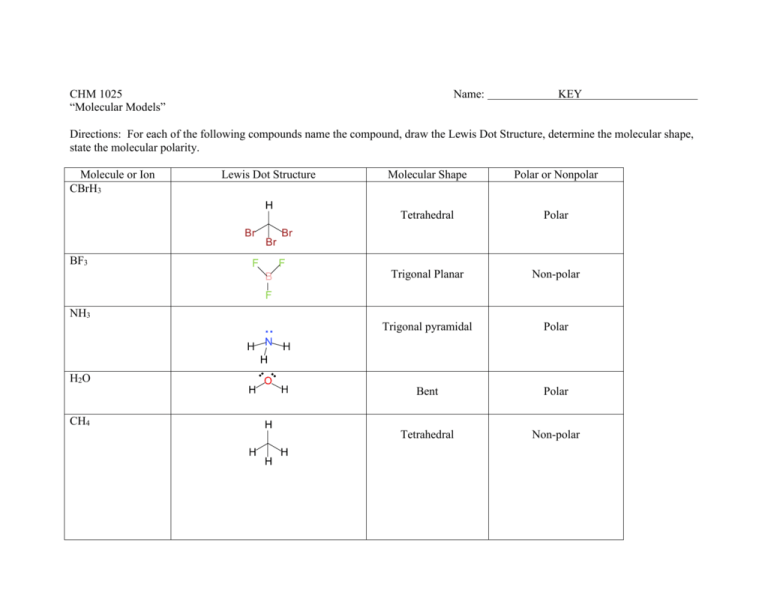
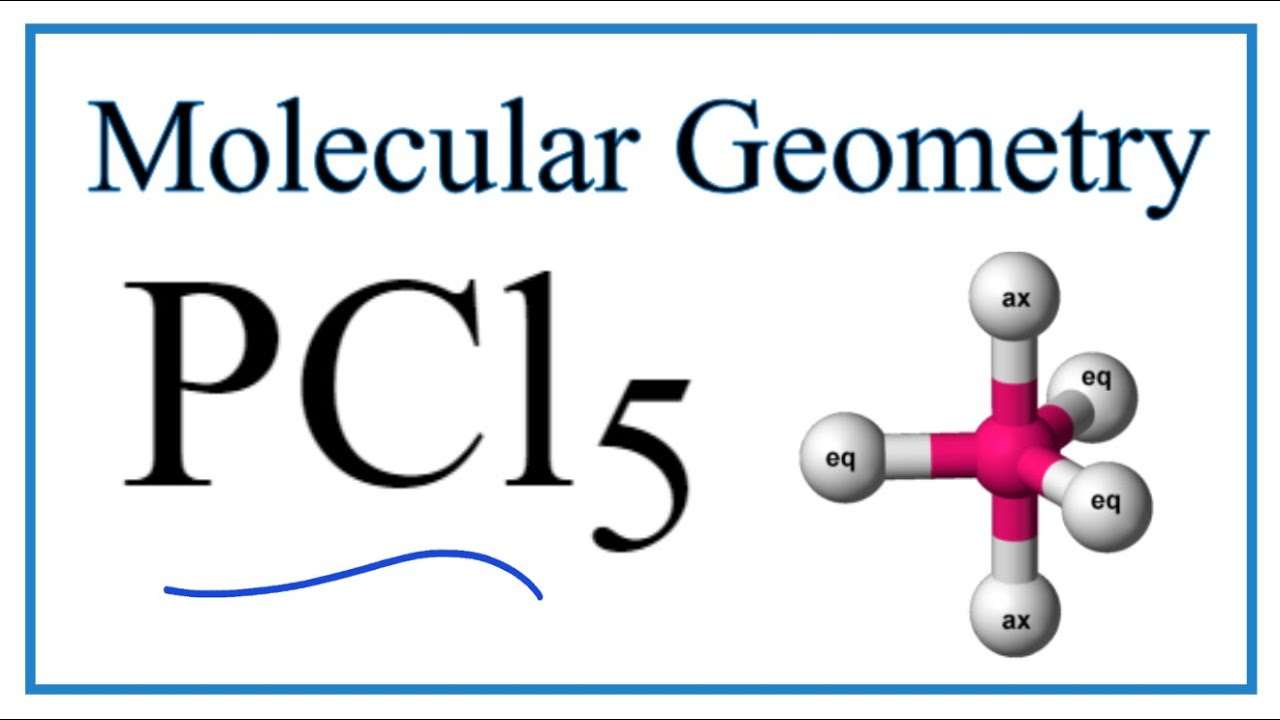
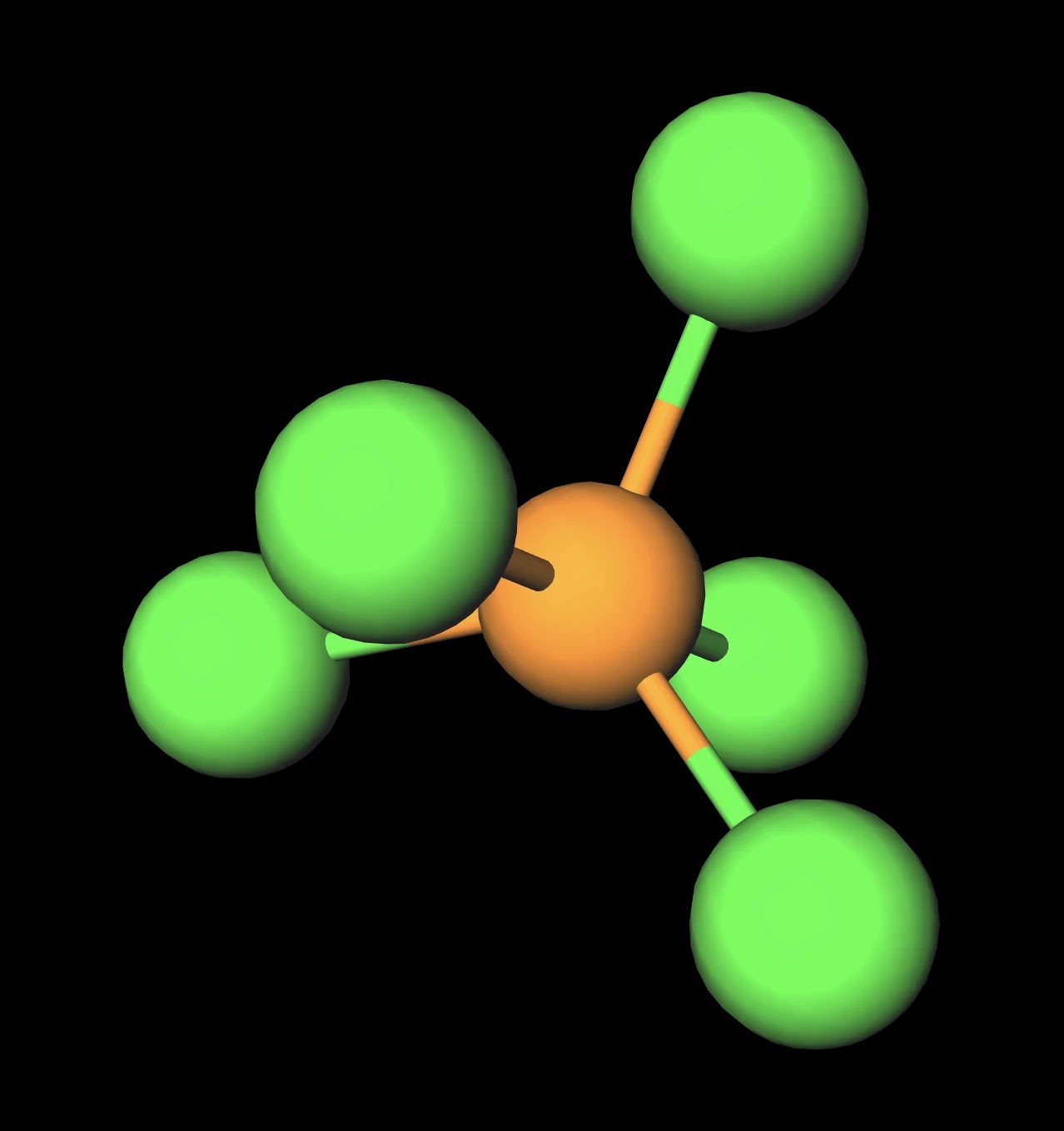
For the molecule PCl5 (phosphorus pentachloride), draw a proper Lewis structure that clearly identifies all electrons and has the predicted geometric (VSEPR) shape. Name and comment on the polarity of the molecule (

What is the Lewis Structure, EN, Bond Polarity, Molecular Geometry and Polarity of Molecule of1. PCi52. - Brainly.ph

PCl5 Lewis Structure, Molecular Structure, Hybridization, Bond Angle, and Shape - Geometry of Molecules