400 ML Solution Of Carbonate (density, Contains 22g Of Sodium Calculate The Mass Per Cent Of Sodium Carbonate In | Sodium Percarbonate 400 Gr | tk.gov.ba

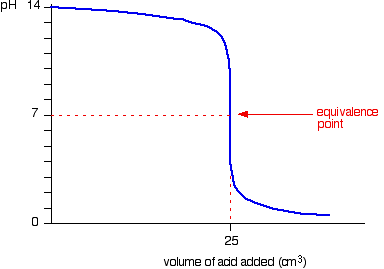
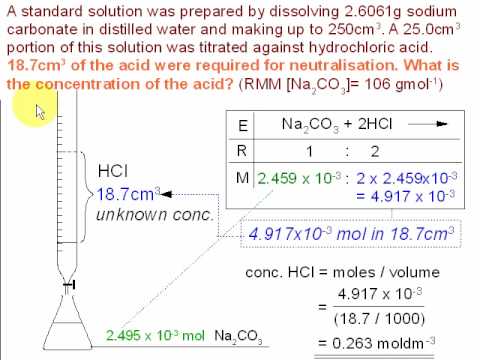
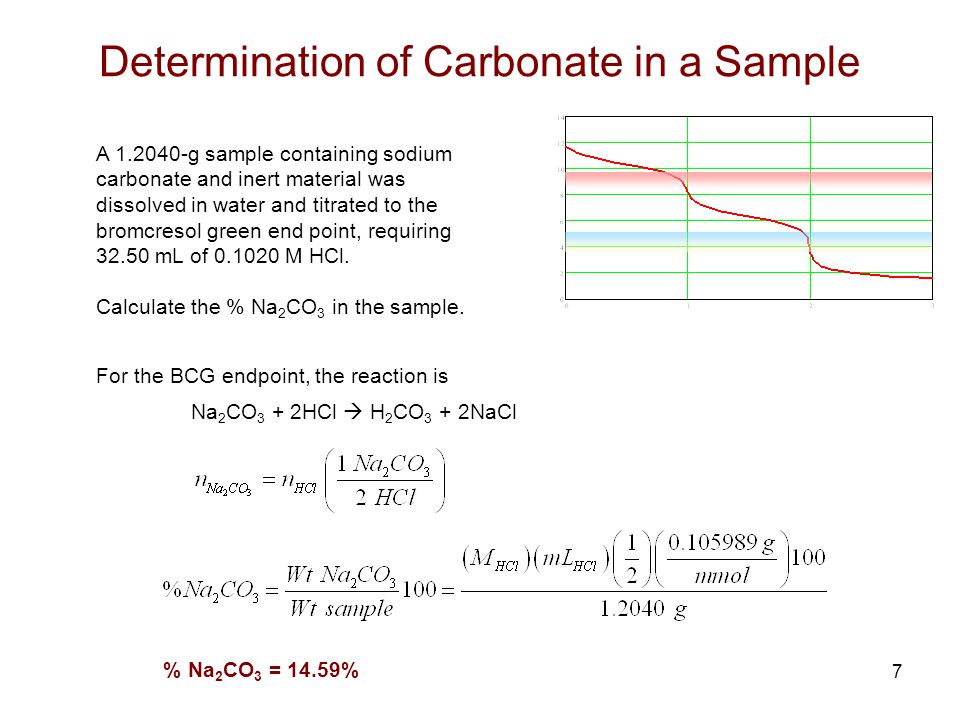
experimental chemistry - Equivalence point of titration of Sodium carbonate - Chemistry Stack Exchange
![Calculate the pH of a buffer solution prepared by dissolving 30g of Na2CO3 in 500 mL of an aqueous solution containing 150 mL of 1M HCl . Ka for: HCO3 = 5.63 × 10^-11 [ log 133150 = - 0.05 ] Calculate the pH of a buffer solution prepared by dissolving 30g of Na2CO3 in 500 mL of an aqueous solution containing 150 mL of 1M HCl . Ka for: HCO3 = 5.63 × 10^-11 [ log 133150 = - 0.05 ]](https://haygot.s3.amazonaws.com/questions/1762025_1744330_ans_4bbe4fbaf6204e3b9a3b783eee4440f1.jpg)
Calculate the pH of a buffer solution prepared by dissolving 30g of Na2CO3 in 500 mL of an aqueous solution containing 150 mL of 1M HCl . Ka for: HCO3 = 5.63 × 10^-11 [ log 133150 = - 0.05 ]

SOLVED: 0.1Mwith the initial pH of O.O1M 50 mL Na2cO3 sodium carbonate solution Calculate the pH values of the Ist and 2nd turning points during the titration with HCI Ka1 1,8.1016 Ka2

APP_H2: Fractional determination for mixture of sodium hydroxide and sodium carbonate|HIRANUMA Co., Ltd.
![50 mL of 0.05 M Na2CO3 is titrated against 0.1 M HCl . On adding 40 mL of HCl , pH of the solution will be[Given for H2CO3, pKa1 = 6.35; pKa2 = 10.33; log 3 = 0.477 , log 2 = 0.30 ] 50 mL of 0.05 M Na2CO3 is titrated against 0.1 M HCl . On adding 40 mL of HCl , pH of the solution will be[Given for H2CO3, pKa1 = 6.35; pKa2 = 10.33; log 3 = 0.477 , log 2 = 0.30 ]](https://i.ytimg.com/vi/8vXKfkDvJjk/maxresdefault.jpg)
50 mL of 0.05 M Na2CO3 is titrated against 0.1 M HCl . On adding 40 mL of HCl , pH of the solution will be[Given for H2CO3, pKa1 = 6.35; pKa2 = 10.33; log 3 = 0.477 , log 2 = 0.30 ]
![SOLVED:A sodium hydrogen carbonate -sodium carbonate buffer is to be prepared with a pH of 9.40 . (a) What must the [HCO3^-] /[CO3^2-] ratio be? (b) How many moles of sodium hydrogen SOLVED:A sodium hydrogen carbonate -sodium carbonate buffer is to be prepared with a pH of 9.40 . (a) What must the [HCO3^-] /[CO3^2-] ratio be? (b) How many moles of sodium hydrogen](https://cdn.numerade.com/previews/ef6e1b45-03cf-464d-9e55-544d05108206_large.jpg)

![PDF] A sodium carbonate-bicarbonate buffer for alkaline phosphatases. | Semantic Scholar PDF] A sodium carbonate-bicarbonate buffer for alkaline phosphatases. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/aaa98c1f9de4c398b6a057bf4936ea76736034c7/1-Table1-1.png)